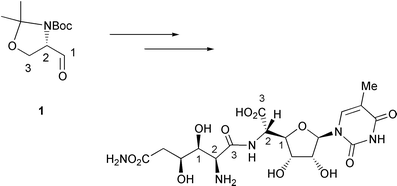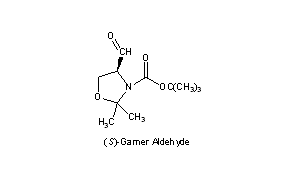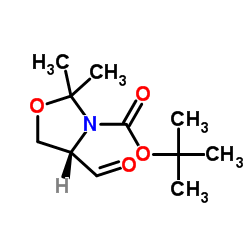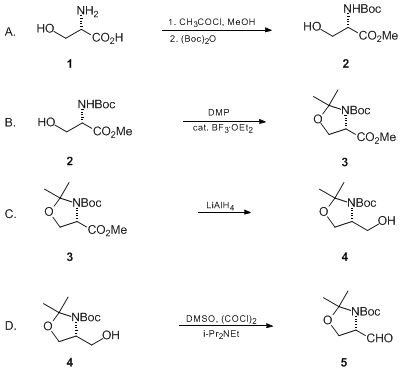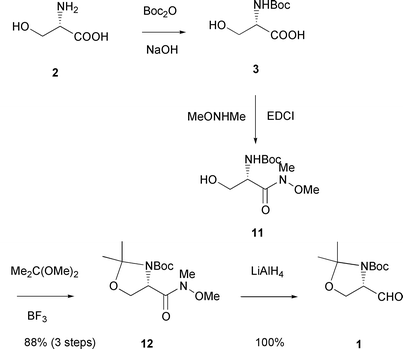
Garner's aldehyde - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/B101054I

PDF) Practical one-step synthesis of ethynylglycine synthon from Garner's aldehyde | Antonella Riu - Academia.edu
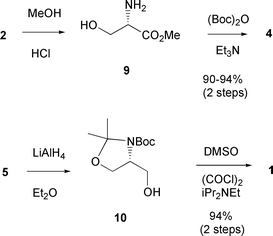
Garner's aldehyde - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/B101054I
![PDF] Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products | Semantic Scholar PDF] Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b3827ac67c2bd364f7c3a3bae47eae474d9a07e9/3-Figure1-1.png)
PDF] Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products | Semantic Scholar

Highly efficient and enantioselective syntheses of (2S,3R)-3-alkyl- and alkenylglutamates from Fmoc-protected Garner's aldehyde | SpringerLink

Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products. - Abstract - Europe PMC
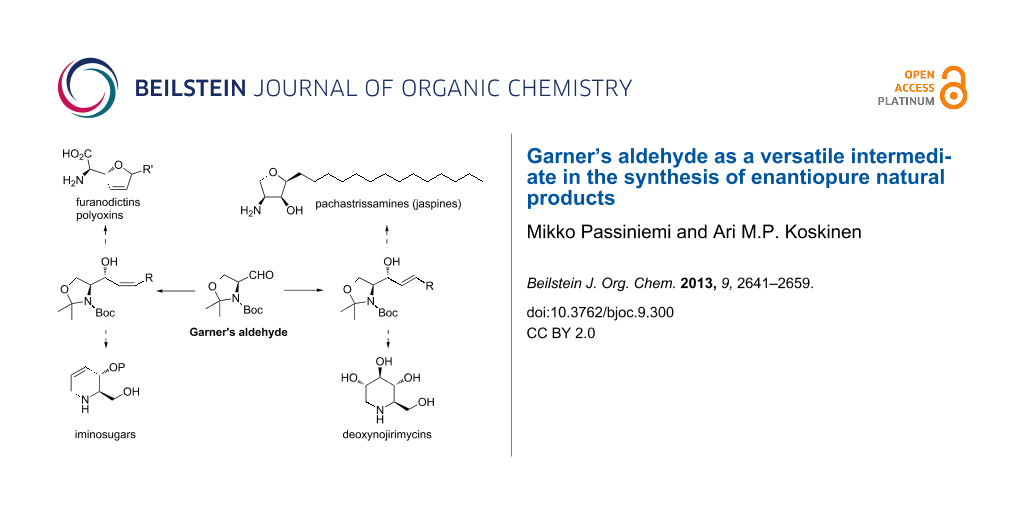
BJOC - Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products

Amazon - Garner's aldehyde: An important chiral α- amino aldehyde in organic chemistry: Ghosh, Moumita: 9786200275783: Books

Stereoselective Synthesis of (2S,6R)‐Diamino‐(5R,7)‐dihydroxy‐heptanoic Acid (DADH): An Unusual Amino Acid from Streptomyces sp. SANK 60404 - Okamura - 2021 - European Journal of Organic Chemistry - Wiley Online Library
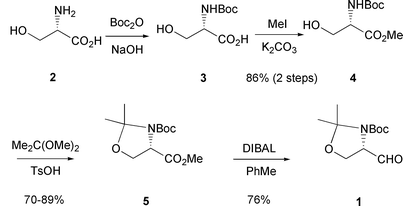
Garner's aldehyde - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/B101054I
![PDF] Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products | Semantic Scholar PDF] Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b3827ac67c2bd364f7c3a3bae47eae474d9a07e9/5-Figure3-1.png)
PDF] Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products | Semantic Scholar

Straightforward Synthesis of Bridged Azaoxa Skeletons: Gold‐Catalyzed Aminoketalization of Garner's Aldehyde‐Derived Alkynes - Alcaide - 2011 - Chemistry – A European Journal - Wiley Online Library

Garner's aldehyde as a versatile intermediate in the synthesis of enantiopure natural products. - Abstract - Europe PMC
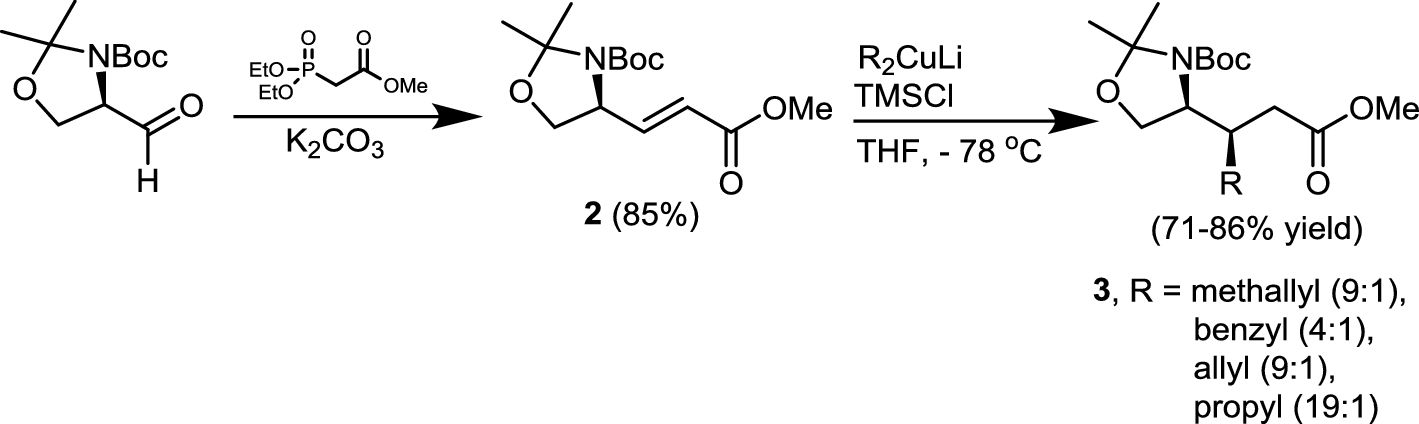
Scheme 1 | Highly efficient and enantioselective syntheses of (2S,3R)-3-alkyl- and alkenylglutamates from Fmoc-protected Garner's aldehyde | SpringerLink